|
We are pursuing to elucidate the mechanism underlying the propagation of prions using yeast prions as a model. The concept of prions has been expanded to include several epigenetically heritable traits in fungi, the yeast prions. Random aggregates are death to cells: diseases. Ablation of the prion protein (PrP) gene in mice prevents scrapie and facilitates production of anti-PrP antibodies. The most common prion diseases of animals are scrapie, which affects sheep and goats, bovine spongiform encephalopathy (BSE) or mad cow disease, and chronic. Amyloids include prions, defined as infectious proteins, which are responsible for mammalian transmissible spongiform encephalopathies such as mad-cow disease. These carbon-rich patches associate strongly with similar patches on other proteins, forming large aggregates. Amyloid aggregates are associated with various human neurodegenerative diseases, including Alzheimer's disease and Huntington's disease. A subset of proteins forms amyloids, which are ordered aggregates composed of cross-beta sheet structures. In addition, we are also working on the biology of prion/amyloids. cerevisiae described above provided the first evidence that chaperone proteins are involved in prion propagation (Chernoff et al., 1995). How chaperones assist in the protein folding in the cell? Which proteins are aggregation-prone? What are the determinants that define the aggregation-prone propensity and the chaperone dependency? In most people, the PrP protein folds normally, leaving the person healthy. These are caused by a certain protein, named PrP, that will stay in a mis-folded conformation (PrPsc) if encouraged to go into it in the first place. Our lab interests are focused on the molecular mechanisms that affect the protein folding in the cell. Diseases caused by prions, like Mad Cow / Creutzfeldt-Jacob are also, in essence, protein folding disorders. Although the folding process is spontaneous, protein folding is often hampered by inter-molecular protein aggregation, which can be prevented by a variety of chaperone proteins in the cell. There is no evidence that the disease is transmitted through. The primary way animals become infected with BSE is by consuming feed contaminated with the infectious BSE agent, an abnormal protein known as a prion. 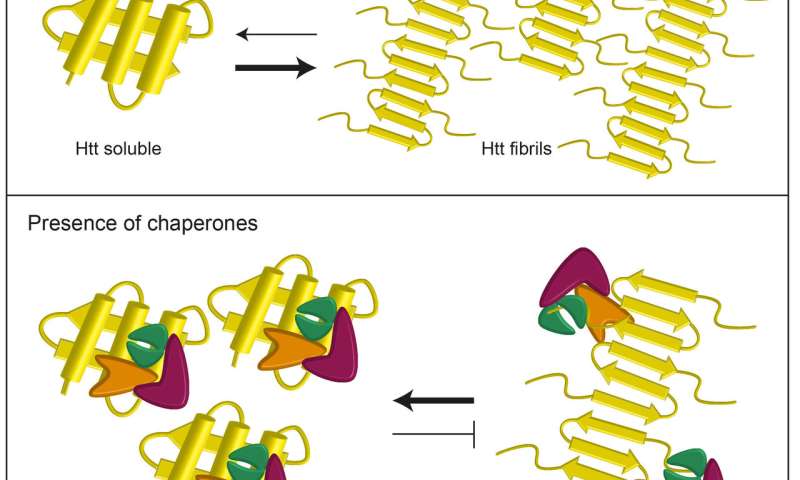
The polypeptide chains, which are emerged from the ribosome in the cell, must fold into the tertiary structures for biological functions. The disease commonly known as 'mad cow' disease is actually bovine spongiform encephalopathy (BSE). Proteins are linear chains of amino acids (polypeptide chains) that adopt unique three-dimensional structures.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
